Optimal Materials for Heat Exchangers Exposed to Hydrochloric Acid (HCl)
Heat Exchanger: A Definition
Heat exchangers are critical components in a range of industrial and commercial systems, their purpose is to transfer heat from one medium to another without the two mediums coming into physical contact. The ‘exchange’ can take place between, liquid-to-liquid, gas-to-liquid or gas-to-gas. Heat exchangers can cool a warm substance or heat a cool substance which is important for systems which need to maintain a steady temperature.
Design and Functionality of a Heat Exchanger
Heat exchangers come in various designs, each tailored for specific needs:
Shell and Tube Heat Exchanger: This type consists of numerous small tubes within a cylindrical shell, where the tubes can be fixed or part of a floating tube stack. The floating design allows for expansion and contraction under different temperatures and easy maintenance.
Plate Heat Exchanger: Operating similarly to shell and tube exchangers, plate exchangers use stacked plates instead of tubes. These are ideal for compact spaces and are common in refrigeration and food and beverage processing.
Air Cooled Heat Exchanger: Often used in mobile applications, these exchangers use air, either from a fan or natural airflow, to cool fluids. They are essential in vehicles or equipment without a steady water cooling source.
Heat Exchanger Efficiency and Industries
Efficiency in a heat exchanger is influenced by the temperature differential, flow rates, and installation. Proper installation and counter-current flow arrangements are crucial for maximising efficiency.
When designing a heat exchanger, there are several key factors to consider for optimal performance:
Temperature Differential: This is the temperature difference between the hot fluid and the coolant. The coolant must be at a consistently lower temperature than the hot fluid. Cooler coolant temperatures are more effective in absorbing heat from the hot fluid, enhancing the heat exchange process.
Flow Rate: The flow rates of fluids on both the primary (hot fluid) and secondary (coolant) sides of the heat exchanger are vital. Higher flow rates can improve the heat transfer capacity of the exchanger. However, they also mean a greater fluid mass, which can complicate the removal of energy and may lead to increased velocity and pressure loss within the system.
Installation Guidelines: Adhering to the manufacturer’s installation guidelines is essential for the efficient operation of a heat exchanger. A common and effective installation method involves a counter-current flow set-up. The coolant and hot fluid flow in opposite directions (e.g. if the coolant flows left to right, the hot fluid should flow right to left).
For shell and tube heat exchangers, it’s recommended that the coolant enters at the lowest inlet position to maintain a full water presence. In air-cooled heat exchangers, proper airflow is crucial; any obstruction in the core can significantly reduce cooling efficiency.
By paying close attention to these aspects, you can ensure your heat exchanger operates at its best, providing effective and efficient heat transfer.

What Industries Use a Heat Exchanger?
Heat exchangers are used across a wide range of industries due to their versatility in managing heat transfer. Some of the key industries include:
- Power Generation: For cooling processes in nuclear, gas and coal-fired power plants.
- Chemical Industry: To control temperatures during chemical reactions and processing.
- Oil and Gas: Utilised in refineries and natural gas processing plants for heating and cooling processes.
- Marine: In ship engines and power plants for cooling systems.
- Automotive: For engine cooling and air conditioning systems.
- Food and Beverage Processing: To heat or cool products and ingredients during manufacturing.
- Pharmaceuticals: Used in the production and formulation of medicines.
- HVAC (Heating, Ventilation, and Air Conditioning): In residential, commercial, and industrial buildings for efficient temperature control.
- Refrigeration: For transferring heat out of an area to cool it, used in industrial, commercial, and domestic refrigeration systems.
- Waste Heat Recovery: To capture and reuse heat from industrial processes.
- Aerospace: For temperature regulation in aircraft systems.
Each of these industries relies on heat exchangers for critical thermal management tasks, highlighting their importance in industrial and commercial applications.

Understanding Failures in a Heat Exchanger
Common issues like oil and water mixing or sediment build-up can often be resolved through proper design, installation and maintenance.
Corrosion from hydrochloric acid can also cause issues, but choosing the correct hydrochloric acid-resistant metals for your application can significantly reduce the risk of damage and extend the lifespan of your equipment.
By selecting materials that are specifically designed to withstand this type of chemical exposure, you can ensure greater reliability and efficiency in your processes, leading to improved safety and cost savings over the long term.
What Is Hydrochloric Acid?
Hydrochloric acid (HCl, also known as muriatic acid) is a strong reducing acid, widely used for pickling steel, making cleaning chemicals and as a starting stock in chemical processing, for instance in the production of vinyl chloride. Although the concentration of HCl used in any application is generally limited to about 38%, it is still considered a highly corrosive solution, especially when metals react with chloride to form metal chlorides.
The presence of metal chlorides and other impurities such as oxidising agents, can drastically increase HCl corrosivity and the dynamic of its corrosive nature. This can lead to rapid corrosion rates, pitting and stress corrosion cracking.
Typical processes that see hydrochloric acid through various concentrations and temperatures include overhead condensing of pharmaceutical and fine chemical products, heating and cooling acids, heat transfer between acids and/or other fluids and acid interchangers etc. These processes make use of highly specialised heat exchangers that are made of suitable materials.
Hydrochloric Acid-Resistant Metals: Hydrochloric Acid (HCl) and Corrosion
Only a few special metals such as titanium (<5% HCI at ambient temperatures only), zirconium and tantalum have a strong passivation tendency and nickel-based and molybdenum-based alloys, with strong thermodynamic stability, can be used in hydrochloric acid medium.
When selecting materials, we must also consider the physical and mechanical properties, economy, supply cycle, working conditions and other factors of the materials for a comprehensive analysis and comparison. For example, Tantalum has the best resistance to hydrochloric acid but suffers from hydrogen embrittlement and inferior mechanical properties as compared to nickel-based alloys.
Hydrochloric Acid-Resistant Metals: Nickel and Nickel-Based Alloys
Nickel (an austenite stabilising element) has good resistance to erosion of non-oxidising acid. The addition of a large amount of nickel can significantly improve the solid solubility of corrosion-resistant alloying elements (such as Cr, Mo, Fe, etc.) and thus form a wide variety of alloys. The passivation element added in the nickel-based alloy can improve the corrosion point and thermodynamic stability of the material.
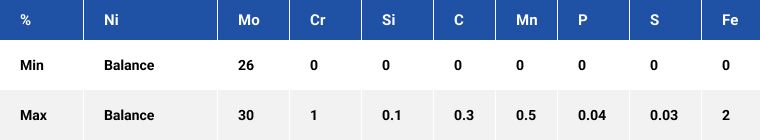
Alloy B-2
A nickel-molybdenum grade alloy which is extremely resistant to hydrochloric acid in a wide range of temperatures. It has excellent corrosion resistance in hydrochloric acid of 160℃ and 2% concentration, the corrosion rate is less than 0.13-0.51 mm/a. It is commonly used in parts of corrosion-resistant equipment under extremely harsh conditions of hydrochloric acid corrosion.
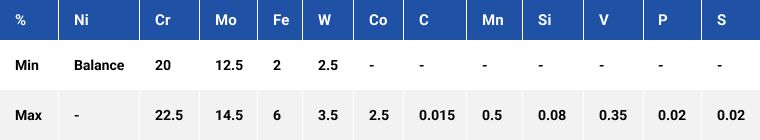
Alloy C276
A solution-annealed nickel-molybdenum-chromium grade alloy with the addition of tungsten. The high molybdenum and chromium contents allow the alloy to perform in oxidising, non-oxidising and mixed acids media, whilst exhibiting outstanding resistance to pitting and crevice corrosion attack. The addition of tungsten inhibits the development of pits.
Industries where C276 is utilised are petrochemical and chemical processing, power generation, pharmaceutical, pulp and paper production and waste treatment to name a few.
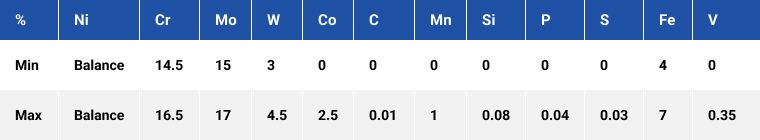
Alloy 22
A nickel-molybdenum-chromium grade with the addition of tungsten. At hydrochloric acid concentrations above about 5%, alloy 22 provides a quantum improvement over the stainless steels and offers much greater resistance to hydrochloric acid than alloy 625 in the concentration range 8 – 20%.
Alloys 200, 400, 625, UNS S32760 and Al-6XN® are known to have good resistance to hydrochloric acid provided that the acid concentrations, temperatures and impurities are closely monitored.
Whether it’s for power plants, marine environments or industrial processes, understanding the types, design and maintenance of heat exchangers is crucial for efficient thermal management. For further information, get in touch.
——————————————————————————————————————————————
AL-6XN® is the registered trademark of Allegheny Ludlum Corporation.
Note that Aluminium, Iron, Copper and their alloys are not suitable for use in any hydrochloric acid applications at any concentrations.
Blogs of Interest
Which Materials are the Best for a Heat Exchanger Application
A Guide to Nickel Alloys: Exploring Properties, Applications and Advantages
Corrosion Resisting Alloys for Aqueous Corrosion Applications Across Industries
FAQs
What is the purpose of heat exchanger?
Heat exchangers play a vital role in transferring heat between two mediums, which can be gases, liquids or a mix of both. In these systems, the mediums are either separated by a solid barrier to avoid mixing or they come into direct contact, depending on the specific requirements of the process.
Is a heat exchanger the same as a boiler?
A boiler operates by heating water through hot gas, and this process is facilitated by a heat exchanger. As the gas heats up, it rises and encounters the heat exchanger, where it is channelled through a coiled pipe, effectively transferring the heat to the water.
Do heat exchangers save money?
One of the most substantial cost-saving advantages of heat exchangers stems from their efficient energy utilisation. In contrast to conventional air conditioning systems that depend on refrigeration mechanisms to cool the air, heat exchangers are designed to eliminate the heat generated by electrical equipment, resulting in significantly lower energy consumption.



